The corollary of adoptive transfer of immunity is that active immunization of BM donors might serve to reduce the incidence of infection in recipients of allo-BMT and that immunization before intensification might likewise be considered in ABMT and ABSCT recipients. Transfer of humoral immunity has been documented from donor to recipient, including immunity to tetanus, varicella, diphteria, influenza virus, cytomegalovirus, hepatitis B virus, and human immunodeficiency virus, 19-23 suggesting that functional B cells are passively transferred by transplantation. 
Because allogeneically transplanted cells can be readily traced, allo-BMT provides direct insight into the origin of B cells in the transplant recipient. Taken together, this suggests that the majority of posttransplantation circulating B cells are poorly differentiated.Īfter ABMT or autologous peripheral blood stem cell transplantation (ABSCT), B cells regenerate from several sources: (1) B cells of the transplant recipient which survived the pretransplantation chemotherapeutic intensification treatment such cells may be seeded in the BM, lymph nodes, or spleen (2) B cells present in the graft (3) hematopoietic stem cell progenitors in the transplant that differentiate after grafting in the recipient and (4) residual recipient stem cells. 12Furthermore, as in neonates, the percentage of mIgM + and mIgD + B cells is high in autologous transplant recipients. During the first year postengraftment, the majority of circulating B cells carry the CD23 +, CD38 + undifferentiated phenotype. CD23 (the low-affinity receptor for the Fc portion of IgE) and CD38 (the ecto-enzyme of nicotinamide (NAD) glycohydrolase that acts as an adhesion/homing receptor and is involved in intracellular calcium homeostasis) are strongly expressed on the circulating B cells of neonates and B cells from cord blood, whereas in adults, CD23 + CD38 +B cells are a minor subset. 11, 12 Thereafter, the numbers of such cells increase to a plateau at 6 to 9 months. 10 The relative and absolute numbers of circulating cells expressing CD19 and CD20, two markers of mature B cells, are decreased during the first 3 months following transplantation. 9 In both its Ig gene rearrangements and in phenotypic expression, B-cell recovery after transplantation appears to recapitulate normal B-cell ontogeny. Normal B-cell differentiation is accompanied by a set of preprogrammed steps of Ig gene rearrangement and by successive acquisition and loss of differentiation level-specific surface molecules. In addition, the potential to accelerate immune reconstitution and the effect that might have in the therapy of malignant disease will be considered. In this review, several strategies that could lead to enhancement of cellular immune function to take particular advantage of posttransplantation minimal residual disease will be discussed. 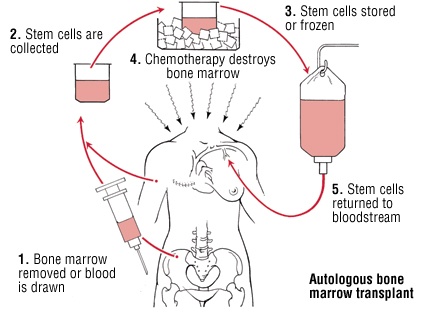
It may have direct clinical implications: Immediately after the administration of intensive cytotoxic drugs, minimal tumor burden is presumed to be present, providing potentially ideal circumstances to eliminate residual disease altogether by immunotherapeutic means. This restoration of immune function is not merely experimental. These include (1) reappearance of functional B cells, (2) thymic and extra-thymic T-cell development, (3) reconstitution of effector cells including cytotoxic T cells and natural killer (NK) cells, and (4) efficient antigen presentation to reconstitute the pretransplantation immune repertoire. Immune reconstitution involves several components of the immune response.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
